 He named it after the asteroid Pallas, which was itself named after the epithet of the Greek goddess Athena, acquired by her when she slew Pallas. In the third period of the table, the atoms all have a neon-like core of 10 electrons, and shell #3 is occupied progressively with eight electrons, starting with the 3s-orbital. In addition, strontium, like rubidium, oxidizes in air and turns a yellow color. Metals are located on the left side of the table, whereas nonmetals are located on the right.The columns are referred to as groups. This is expressed by the law of periodicity." Dmitri Mendeleev, Principles of Chemistry, Vol. 6 elements = Li, Na, K, Rb, Cs, Fr, Click on the group number for the Alkali Metals family at the top of the table. Answer and Explanation: 1 Become a member to unlock this answer!, Alkali metals. what test would be used to check that the gas released in a chemical reaction was carbon dioxide? Which element in period 5 is the most active metal? Iodine is a chemical element with the symbol I and atomic number 53. The process of moving from one open window to another is called what? You can specify conditions of storing and accessing cookies in your browser, What is the element in group 10 and period 5. 5 or 6 elements F, Cl, Br, I, At, Uus (possibly), Click on the group number for the Halogen family at the top of the table. These superconducting alloys, also containing titanium and tin, are widely used in the superconducting magnets of MRI scanners. The position of elements in the modern periodic table is determined by determining periods and groups in the periodic table. It is also possible to analyze the ionization energy, electromagnetism, electron affinity, and so on. Interest in the metal grew after Wood's findings were reported to the Royal Society. 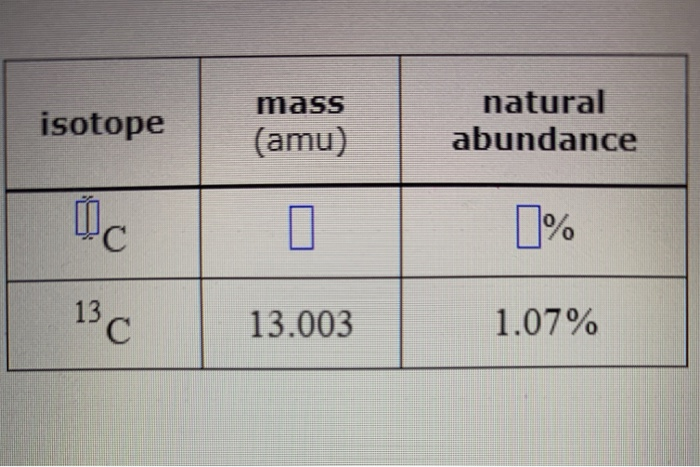
Like zinc, it prefers oxidation state +2 in most of its compounds and like mercury it shows a low melting point compared to transition metals. The element in group 1 and period 7 = Fr 4. The element in group 18 and period 6= Rn e. The element in group 2 and period 3 = Mg d. The element in group 15 and period 4 = As c. List the member elements by The element in group 10 and period 5 = Palladium b. If it does come in contact with water, though, it will combine with the atoms of both oxygen and hydrogen to form strontium hydroxide and pure hydrogen gas which quickly diffuses in the air. "If all the elements are arranged in the order of their atomic weights, a periodic repetition of properties is obtained. The Group 7 elements are called the halogens. members are generally used as conductors of electricity. Finally, all the metals in group 14 have a +4 charge. Thus, The element in group 10 and period 5 is palladium or Pd. Can you see a pattern for their electron numbers? b. According to Madelung's rule, the electronic configuration of palladium and platinum are expected to be 5s2 4d8 and 4f14 6d2 5d8 respectively. 
20.9032.Indium has no biological role and can be toxic as well as Antimony. That'S our first question: what's our second question antimony, so this one's about antimony, it's got 2 naturally occurring isototes. It can't be 94 percent that and get this can't be 94 percent f e 57 with 55.845 atomic mass. So yes, it is likely to have other isototes. 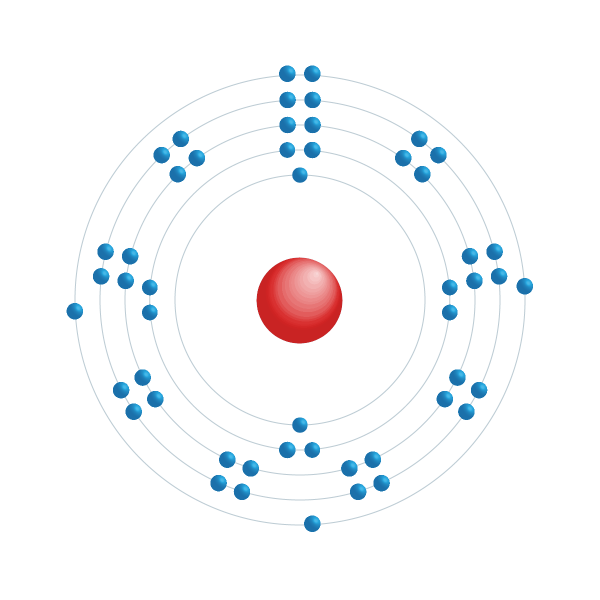
There must be other isotopes likely iron 55 to calculate the known atomic mass. The molar mass of iron is 55.845 mumsince. Am us and a percentage abundance of 56.935 opson random roinnumber tare of unknown? Is it likely to have other eyes likely to have other isototesis it likely to have other isotopes ye? Okay? Is it likely to have at isototes? So let me see here i just had to take a drink coffee. Am us, and a relative abundance of 5.845 are natural percent abundance as given right there. We'Re told that high iron has at least 2 isototes and were given the following information: iron 54, which has a mass of 53.9396 joule. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
